FAQ – Frequently Asked Questions
Questions about flow chemistry and microreactors in general
Q: What is flow chemistry?
A: Flow chemistry is the general term used for continuous operation of chemical reactions. In bulk industrial chemical processes, flow chemistry has been in use for a long time, but in chemical laboratories and smaller scale manufacturing, it is a relatively new phenomenon. The introduction of flow chemistry in the lab environment is mainly due the development of microreactors.
Q: What is a microreactor?
A: Microreactors are devices in which mixing and/or chemical reactions take place. Inside microreactors, small channels and other flow guiding geometries are present, generally having small lateral dimensions (<1mm), resulting in an improved temperature control and reduced mass transfer limitations. Therefore, microreactors are ideal for fast and exothermic chemical reactions.
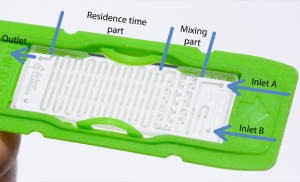
Q: What are main advantages of using flow chemistry?
A: Flow chemistry offers a new set of tools for the synthetic chemist, being complementary to conventional batch techniques. Benefits and advantages include:
• Improved control over process parameters in a straightforward fashion.
• A new range of process conditions available: sub second reaction times, easy operation above solvent boiling point.
• Enhanced safety for production of chemical compounds
• Less chemical waste and less consumption of precious starting compounds
Q: But I can do flow chemistry with simple tubing, why use microreactors?
A: Microreactors offer several benefits over ‘tubing’ flow chemistry: the volume is very small, saving precious chemical compounds during optimisation. Special structures such as mixing geometries offer highly improved mixing performance. Furthermore, temperature control is much easier using microreactors.
Questions about flow chemistry’s suitability for your application
Q: What are the main requirements for a chemical process to be suitable for Microreactors?
A: The two main requirements are full dissolution and speed: no solid particles must be present in the reaction mixture, and we recommend a maximum reaction time of 20 minutes for FlowStart FlowStart Evo.
Q: Many reaction, such as e.g. the Clemmenson reduction, require solid particles. Are your microreactors suitable for such reactions? And is precipitation a problem for your microreactors?
A: Flow chemistry in general is mostly performed in solutions without any solid particles, because particles can cause clogging (fouling) of all parts of the system, in particular of the reactor. However, in some cases the reactor can handle very small particles during the quench phase. In some cases flowchemistry is used for producing of solid particles such as nano particles
Q: Do I have to quench the reaction? And how?
A: That is certainly possible with for example the Gas Module. Please contact us to discuss about regulating the gas flows and what infrastructure is already in place at your location.
Q: Which microreactor do I need for my reaction?
A: We have a wide range of standard micoreactors available, covering most applications. However, if your application is not covered, we can design any reactor for your specific needs.
Q: Where can I find literature references?
A: You can find a list of references in our section for patents and publications.
Q: Can I use flow chemistry for biocatalysis (enzymatic reactions)?
A: Yes, enzymatic reactions can be carried out with flow chemistry. For example, the enzymatic cyanohydrins formation is known to work excellently in our microreactors.
For more detailed information see: http://dx.doi.org/10.1002/bit.21649
Q: My chemical reaction is not in the list. How do I know that my chemical process in suited for flow chemistry?
A: Simply by getting in contact with us, so we can help you further.
Q:1. Some chemical reactions produce gasses as a by-product. Is the FlowStart Evo suited for such reactions?
A: If gasses emerge from solution, the effective reactor volume is reduced, resulting in a decreased total reaction time accordingly. Although this is not always a problem, it can be prevented by increasing the back pressure of the complete system by using a back pressure regulator.

Questions about how to use flow chemistry
Q: How do I translate conventional batch chemistry into micro/macro flow chemistry?
A: Generally, this is the most difficult and time-consuming phase in implementing flow chemistry. FutureChemistry has a combined flow chemistry experience of over 10 years, and is happy to assist you in this process.
Q: Do I have to quench the reaction? And how?
A: That is certainly possible with for example the Gas Module. Please contact us to discuss about regulating the gas flows and what infrastructure is already in place at your location.
Q: How do I analyze product obtained with flow chemistry?
A: Any conventional analysis technique can be used. The product flow is collected in any vessel of choice, after which the mixture is worked up and analysed as required for you specific reaction. Currently we are developing an integrated phase separation utility. Please subscribe for the newsletter to stay updated about this and other developments.
Q: How easy can Microreactor Technology be used for scaling up?
A: It is generally known that scaling up using conventional methods is a laborious and time-consuming process. We can ensure you that by using our microreactor technology you can drastically decrease this process in time.
Still, scaling up chemical processes is not easy. For this we collaborate with other companies unified in the consortium Access2Flow. In this way we can guarantee the best support and guidance for the translation from traditional laboratory batch synthesis, to full continuous flow processes both on micro and macro-scale.
Questions about our products
Q: Is the FlowStart Evo also available for rent?
A: We do have systems available for rent, under certain conditions. Please contact us to discuss rental and your application.
Q: Can I also connect two or multiple FlowStarts in order to conduct multistep reactions under various conditions?
A: Sure, again this is one of the advantages our microreactor technology has to offer. An alternative would be to use a special microreactor with multiple reaction stages.
